If you or your patients have ever noticed that a stressful week at work somehow ends up written across the skin the sudden flare of redness, the maddening itch, the restless nights you are not imagining it. That connection is real, and until very recently, medicine could only describe it in vague terms: “stress weakens the immune system,” “stress disrupts the skin barrier.” True enough, but profoundly incomplete.
A landmark study published in Science on 19 March 2026 (Tian et al., DOI: 10.1126/science.adv5974) has changed that entirely. For the first time, researchers from Fudan University have mapped the precise, cell-by-cell, molecule-by-molecule pathway through which psychological stress activates the skin’s immune system and the findings carry major implications for how we understand and manage atopic dermatitis (AD), eczema, and related inflammatory skin conditions. This article breaks down the science, contextualises it within the broader literature on the neuroimmune and gut–skin axes, and explores what it means in practice.
🔬 Core Finding at a Glance
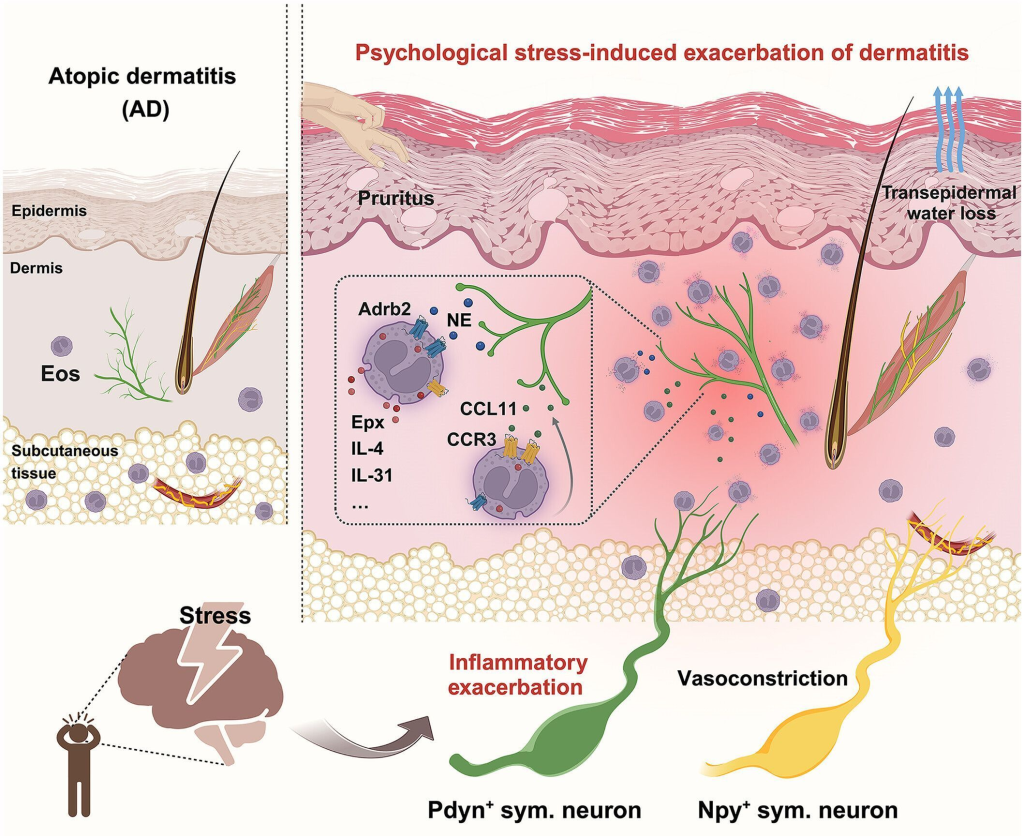
A specific subset of prodynorphin-positive (Pdyn⁺) noradrenergic sympathetic neurons that directly innervate hairy skin act as the neural bridge between the brain’s stress circuits and the skin’s immune response recruiting and activating eosinophils to drive inflammation in a stress-dependent manner.
What the Study Actually Found: A New Neural – Immune Circuit
The research team began by asking a deceptively simple clinical question: do patients with higher stress levels show more severe atopic dermatitis? Analysing data from 51 human patients with AD, they confirmed that higher self-reported stress correlated with both greater eosinophil accumulation in skin biopsies and more severe clinical inflammation. This was not new in itself the stress-eczema association has been observed for decades. What came next, however, was groundbreaking.
Using mouse models engineered to replicate AD-like skin inflammation, the researchers exposed subjects to psychosocial stressors and tracked the resulting cellular cascade in exquisite detail. The key discovery was the identification of a distinct subpopulation of sympathetic neurons that preferentially innervate hairy skin: the Pdyn⁺ (prodynorphin-positive) noradrenergic neurons. Unlike the more widely studied Npy⁺ sympathetic neurons, this subset acts as a dedicated stress-to-skin signalling line.
When stress is perceived, these Pdyn⁺ neurons transmit signals from the brain’s stress-response centres directly to the skin. The mechanism proceeds as follows:
The Stress → Skin Inflammation Cascade (Tian et al., 2026)
- Brain perceives stress → activates the central sympathetic stress-response network.
- Pdyn⁺ sympathetic neurons in hairy skin receive the signal and release CCL11 (eotaxin-1), a chemokine that acts as a recruitment signal for eosinophils.
- CCL11 binds CCR3 receptors on circulating eosinophils → eosinophils are attracted to the inflamed skin tissue in large numbers.
- Neurons simultaneously release norepinephrine (NE), which activates eosinophils via beta-2 adrenergic receptors (Adrb2).
- Activated eosinophils discharge cytotoxic granule proteins (e.g., eosinophil peroxidase, Epx) and proinflammatory cytokines, notably IL-31 — a key driver of itch.
- Local inflammation amplifies, worsening barrier dysfunction, redness, and the itch–scratch cycle characteristic of AD.
When the team used optogenetic tools to artificially activate these Pdyn⁺ neurons, the proportion of eosinophils in the skin more than doubled. Conversely, when these neurons were genetically ablated, or when eosinophils were eliminated, stress no longer worsened inflammation. The eczema itself remained driven by other mechanisms but the stress-induced exacerbation was entirely abolished. Both components of the axis are necessary for the stress effect.
“Our findings reveal a neuroimmunological mechanism underlying psychological stress–induced exacerbation of dermatitis, emphasising the Pdyn⁺ sympathetic–eosinophil axis as a crucial interface between the brain and skin inflammation, with potential therapeutic implications.” Tian et al., Science, 2026
Why This Matters: The Bigger Picture of Neuroimmune Crosstalk
This discovery does not stand alone. It slots into a rapidly growing body of literature recognising that the skin is not a passive target of immune dysfunction but an active neuroimmune organ, intimately connected to the brain, the gut, and the endocrine system.
The HPA Axis and Chronic Stress
Until now, much of the focus on stress and skin disease centred on the hypothalamic–pituitary–adrenal (HPA) axis the classical stress hormone pathway. Chronic stress elevates cortisol, which can compromise skin barrier function and paradoxically worsen inflammation despite cortisol’s anti-inflammatory reputation in acute settings. Research published in Brain, Behaviour, and Immunity (Zhang et al., 2024) confirmed that neuroendocrine–immune interactions are central to the pathogenesis of stress-associated skin diseases, with cortisol signalling creating a cycle of barrier dysfunction and immune dysregulation.
Tian et al.’s work adds a critical new layer: alongside the HPA axis, there exists a direct sympathetic neural highway to the skin that can act far more rapidly than hormonal signalling — transmitting stress-to-inflammation commands in real time, neuron by neuron.
A Parallel Detrimental Cycle: SNS Dysfunction in AD
Complementary evidence published in the Journal of Allergy and Clinical Immunology: In Practice (2025) found that patients with AD show measurable sympathetic nervous system (SNS) dysfunction including diminished perspiration and attenuated electrodermal responses alongside compensatory hypertrophy of the sympathetic ganglia. Chemogenetic inhibition of sympathetic neurons in mouse models worsened inflammation, whilst targeted activation attenuated it. This reveals a reciprocal, damaging cycle: AD inflames and damages sympathetic nerve function, and that dysfunction in turn exacerbates the disease.
The Gut–Skin-Brain Axis: Microbiota as the Missing Link
For those of us working at the intersection of clinical nutrition, neurogastroenterology, and skin health, the Tian et al. findings take on an additional dimension when considered alongside the emerging science of the gut–skin axis.
Atopic dermatitis is increasingly framed not as a purely cutaneous condition, but as a multisystem disorder involving immune dysregulation, gut microbial dysbiosis, neuroendocrine imbalance, and epidermal barrier dysfunction — an interconnected network some researchers have termed the SINGE system (Skin–Immune–Neuro-Gastro-Endocrine; Paz & Lio, Dermatology Practical & Conceptual, 2025).
| Axis / System | Key Mechanism | Implication in AD |
|---|---|---|
| Sympathetic–Eosinophil (NEW) | Pdyn⁺ neurons → CCL11/CCR3 recruitment + NE/Adrb2 activation of eosinophils | Direct neural driver of stress-induced flare-ups |
| HPA Axis | Cortisol → compromised barrier, immune dysregulation | Chronic stress amplifies inflammation via hormonal route |
| Gut–Skin Axis | Gut dysbiosis → reduced SCFAs → impaired immune tolerance, disrupted skin barrier | Gut microbiome shapes systemic immune tone relevant to AD |
| Neuro-Gut–Skin Loop | Stress → SNS activation → gut barrier permeability → systemic inflammation → skin | Stress simultaneously fires the skin and destabilises the gut |
A narrative review in the Journal of Applied Physiology (2024) confirmed that psychosocial stress activates both the SNS and HPA axis, causing redistribution of immune cells, elevation of proinflammatory cytokines (IL-1, IL-6, TNF-α), and — critically — disruption of the gut microbiome composition and increased intestinal permeability. This gut dysbiosis in turn stimulates further HPA and SNS activation, creating a positive feedback loop driving chronic proinflammatory states.
The gut microbiota modulates the skin’s immune environment through several pathways: short-chain fatty acids (SCFAs) such as butyrate regulate T-cell tolerance and suppress Th2-skewed inflammation; tryptophan metabolites influence mucosal immunity; and bacterial signals calibrate systemic cytokine tone. A systematic review published in Gut Microbes (2024) identified that probiotics — particularly multistrain Lactobacillus-dominant formulations combined with Bifidobacterium — show modest but consistent reductions in AD severity scores and pruritus in paediatric populations, with effects mediated in part through restoration of microbial diversity and SCFA production.
Early-onset persistent AD has also been associated with reduced intestinal acetic acid levels and altered abundance of Ruminococcus gnavus — findings that directly challenge a skin-only view of the disease and underscore the importance of gut-targeted interventions (Barman et al., 2024, as cited in Frontiers in Microbiology, 2025).
Clinical Implications: What Can We Do With This Knowledge?
The Tian et al. study is mechanistic and primarily preclinical, but its translational implications are substantial. As Nicolas Gaudenzio and Lilian Basso noted in an accompanying Perspective in Science, this work offers a mechanistic explanation for what clinicians and patients have observed for decades — and opens the door to highly targeted therapeutic strategies.

1. Targeting the Neural Pathway
Blocking the Pdyn⁺ sympathetic neurons or their downstream signals (CCL11–CCR3 axis; Adrb2 receptor on eosinophils) represents a genuinely novel class of therapeutic target. Beta-2 adrenergic receptor modulation is already explored in respiratory medicine; its relevance to skin neuroimmunology is now scientifically grounded. Anti-eosinophil biologics such as benralizumab have shown early promise in AD (Whetstone et al., Clinical and Translational Allergy, 2025), and the current study strengthens the rationale for targeting this cellular population in stress-sensitive AD phenotypes.
2. Stress Management as a Clinical Tool – Not a Soft Add-on
Perhaps most importantly for everyday clinical practice, this study validates stress management as a mechanistically grounded intervention — not merely a lifestyle recommendation. Reducing sympathetic nervous system hyperactivation through evidence-based approaches directly interrupts the Pdyn⁺ neuron → eosinophil cascade. Relevant modalities include:
- Mindfulness-based stress reduction (MBSR) demonstrated to reduce SNS activity and lower circulating inflammatory cytokines.
- Biofeedback and heart rate variability training directly improves vagal tone and modulates sympathetic output.
- Sleep optimisation disrupted sleep elevates sympathetic activity and IL-31, a key cytokine in the Tian et al. pathway.
- Adaptogens and magnesium growing evidence for modulating the HPA axis response, though always within an integrative, clinician-supervised framework.
3. Nutritional and Microbiome-Centred Strategies
Given the convergent evidence from the gut–skin axis literature, a microbiome-informed nutritional approach remains one of the most powerful and accessible interventions in our clinical toolkit. When the gut microbiome is dysbiotic, systemic immune dysregulation is amplified — feeding directly into the inflammatory state that the newly identified sympathetic–eosinophil axis relies upon. Practical strategies include:
- Prebiotic fibre (inulin, FOS, diverse plant polyphenols) to support SCFA-producing communities, notably Faecalibacterium prausnitzii and Akkermansia muciniphila.
- Targeted probiotic supplementation — multistrain formulations including Lactobacillus rhamnosus GG, L. plantarum, and Bifidobacterium longum with the strongest evidence base in AD.
- Anti-inflammatory dietary patterns — Mediterranean-style diets rich in omega-3 fatty acids (EPA/DHA), polyphenols, and fermented foods reduce Th2 inflammatory skewing.
- Tryptophan-rich foods — support serotonin and kynurenine pathways with downstream effects on gut barrier integrity and immune tone.
- Avoiding inflammatory triggers — ultra-processed foods, excess refined sugars, and dietary emulsifiers that drive intestinal permeability and worsen gut dysbiosis.
🌿 Want a Personalised Gut–Skin-Stress Protocol?
Our clinical team at Smart Nutrition International specialises in neurogastroenterology, microbiome assessment, and integrative skin health. Book a consultation and receive a fully personalised programme.Book a Clinical Consultation
Looking Ahead: Open Questions and Future Research Directions
As Gaudenzio and Basso noted in their accompanying Perspective, the Tian et al. findings raise important questions that will drive the next wave of research. Do similar sympathetic–eosinophil mechanisms operate in other stress-sensitive inflammatory diseases such as psoriasis, inflammatory bowel disease, or rosacea? The skin shares its neuroimmune architecture with the gut mucosa in several respects — the sympathetic innervation of the intestinal wall and its immune modulation is a well-established but still evolving field.
Equally, the current study was conducted in murine models, with human evidence limited to correlational data from 51 patients. Larger human trials, including skin biopsy studies correlating Pdyn⁺ neuronal density with AD phenotype and stress biomarkers, are now urgently needed. The development of selective CCL11/CCR3 antagonists or topical Adrb2 modulators with a skin-specific mechanism would represent a genuinely new therapeutic class.
For clinicians in integrative and nutritional medicine, the most exciting implication is conceptual: this research confirms that the brain, the gut, and the skin are not three separate systems requiring three separate specialists. They are one integrated neuroimmune organism, and caring for the whole – through stress management, microbiome nutrition, and targeted supplementation — is not complementary medicine. It is, increasingly, the science.
Summary: Key Clinical Takeaways
- A specific subset of Pdyn⁺ sympathetic neurons connecting the brain to hairy skin mediates stress-induced atopic dermatitis flare-ups via eosinophil recruitment and activation.
- The signalling cascade involves CCL11→CCR3 (recruitment) and norepinephrine→Adrb2 (activation), culminating in IL-31 release and amplified skin inflammation.
- Both the HPA axis (cortisol) and the direct sympathetic–eosinophil axis are active in stress-related AD — they are complementary, not competing, mechanisms.
- Gut microbiome dysbiosis worsens the systemic inflammatory background that this neural axis fires into — making nutritional and microbiome strategies mechanistically justified.
- Stress management, targeted probiotic nutrition, and anti-inflammatory dietary patterns are now supported by mechanistic, not merely observational, evidence in AD care.
Scientific References
- Tian J, Cao Y, Li Y, et al. A sympathetic-eosinophil axis orchestrates psychological stress to exacerbate skin inflammation. Science. 2026;391(6791):1269–1277. doi:10.1126/science.adv5974
- Gaudenzio N, Basso L. [Perspective] Stress and skin — a neural–immune connection decoded. Science. 2026;391(6791). Accompanying Perspective to Tian et al.
- Zhang H, Wang M, Zhao X, et al. Role of stress in skin diseases: a neuroendocrine-immune interaction view. Brain, Behav Immun. 2024;116:286–302.
- [SNS dysfunction study] Sympathetic nerve dysfunction exacerbates skin inflammation in atopic dermatitis. J Allergy Clin Immunol Pract. 2025;13(11):2927–2935. ScienceDirect
- Paz M, Lio P. Skin-Immune-Neuro-Gastro-Endocrine (SINGE) system: lighting the fire on atopic dermatitis research. Dermatol Pract Concept. 2025;15(4).
- Wrześniewska M, Wołoszczak J, Świrkosz G, Szyller H, Gomułka K. The role of the microbiota in the pathogenesis and treatment of atopic dermatitis — a literature review. Int J Mol Sci. 2024;25(12):6539.
- [Gut–skin axis probiotics review] From gut dysbiosis to skin inflammation in atopic dermatitis: probiotics and the gut–skin axis. Int J Mol Sci. 2027;27(1):365. MDPI
- Salem I, Ramser A, Isham N, Ghannoum MA. The gut microbiome as a major regulator of the gut-skin axis. Front Microbiol. 2018;9:1459.
- [Gut–skin axis bidirectional review] The gut-skin axis: a bi-directional, microbiota-driven relationship with therapeutic potential. Gut Microbes. 2025;17:2473524.
- [Psychosocial stress–gut microbiome review] Exploring the complex relationship between psychosocial stress and the gut microbiome: implications for inflammation and immune modulation. J Appl Physiol. 2024. doi link
- Whetstone CE, et al. Benralizumab depletes IL-5Rα-bearing cells in skin lesions of patients with atopic dermatitis. Clin Transl Allergy. 2025;15:e70090.
- [Frontiers Microbiology bibliometric] Current insights and trends in atopic dermatitis and microbiota interactions: a systematic review and bibliometric analysis. Front Microbiol. 2025. doi:10.3389/fmicb.2025.1613315

