Understanding the Impact on Parietal Cells, Microbiota, and Nutritional Status
Introduction: Beyond Respiratory Symptoms COVID-19’s Gastrointestinal Legacy
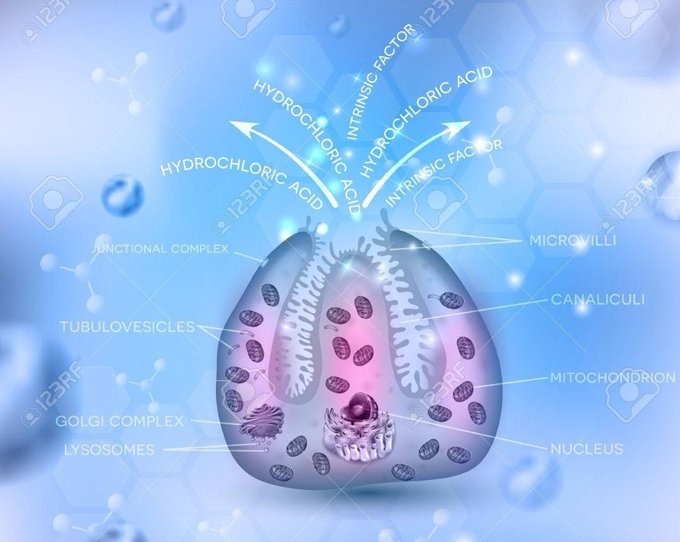
Since the emergence of SARS-CoV-2 in late 2019, our understanding of COVID-19 has evolved dramatically. Whilst initial attention focused primarily on respiratory manifestations, mounting evidence reveals that this novel coronavirus possesses a uniquely broad tissue tropism, affecting multiple organ systems far beyond the lungs. Among the most clinically significant yet underappreciated consequences is the virus’s destructive impact on gastric parietal cells—the specialised epithelial cells responsible for secreting hydrochloric acid and intrinsic factor within the stomach lining.
Recent research published in Frontiers in Microbiology and PubMed has illuminated a concerning mechanism: SARS-CoV-2 directly damages parietal cells in the gastric mucosa, compromising the stomach’s protective acid barrier and disrupting the synthesis of intrinsic factor, a glycoprotein essential for vitamin B12 absorption. This gastric dysfunction creates a cascade of consequences, including increased susceptibility to pathogenic bacterial colonisation (notably Helicobacter pylori), gastric dysbiosis, and nutritional deficiencies that can persist long after the acute infection resolves.
This article examines the scientific evidence behind COVID-19’s gastric impact, explores the nutritional and microbiological implications, and provides evidence-based guidance for clinical dietitians, healthcare professionals, and patients navigating post-COVID recovery.
The Unique Pathophysiology: Why COVID-19 Differs from Influenza
Viral Tropism and ACE2 Receptor Expression
SARS-CoV-2 gains cellular entry primarily through the angiotensin-converting enzyme 2 (ACE2) receptor, which is abundantly expressed not only in respiratory epithelium but also throughout the gastrointestinal tract, including gastric parietal cells. This widespread receptor distribution enables the virus to infect diverse tissues, distinguishing it fundamentally from seasonal influenza viruses, which predominantly target respiratory epithelium.
Research demonstrates that ACE2 expression in gastric tissue provides SARS-CoV-2 with a direct pathway to invade and damage parietal cells. In contrast, influenza viruses lack this gastric tropism and do not systematically compromise gastric acid production or intrinsic factor synthesis. This distinction is critical: COVID-19 is not simply “another flu” its multisystem pathology reflects fundamentally different viral behaviour and tissue targeting.
Parietal Cell Destruction and Functional Consequences
Parietal cells serve two vital physiological functions:
- Hydrochloric acid (HCl) secretion: Maintains gastric pH between 1.5-3.5, creating a hostile environment for ingested pathogens
- Intrinsic factor production: Binds dietary vitamin B12, enabling its absorption in the terminal ileum
When SARS-CoV-2 infects and destroys these cells, both functions are compromised. Studies cited in the Frontiers in Microbiology article reveal that COVID-19 patients exhibit reduced gastric acidity (hypochlorhydria) and diminished intrinsic factor levels during and following acute infection. This cellular damage may persist for months, contributing to the constellation of symptoms recognised as Long COVID or post-acute sequelae of SARS-CoV-2 infection (PASC).
Clinical Consequences: Infection Risk and Microbiome Disruption
Compromised Gastric Acid Barrier
The stomach’s acidic environment serves as the body’s first chemical defence against ingested pathogens. Gastric pH below 3.0 effectively neutralises most bacteria, viruses, and parasites consumed in food and water. However, when parietal cell function is impaired and gastric pH rises (becomes less acidic), this protective barrier weakens significantly.
Increased susceptibility to Helicobacter pylori: This spiral-shaped bacterium thrives in conditions of reduced gastric acidity. Whilst H. pylori possesses urease enzymes that enable survival in acidic environments, colonisation becomes substantially easier when gastric pH is elevated. Given that H. pylori infection is associated with chronic gastritis, peptic ulcers, and increased gastric cancer risk, the post-COVID vulnerability to colonisation represents a significant public health concern.
Small intestinal bacterial overgrowth (SIBO): Reduced gastric acidity also permits abnormal bacterial migration from the colon into the small intestine, where bacterial populations should normally remain minimal. SIBO manifests with bloating, diarrhoea, malabsorption, and nutritional deficiencies—symptoms frequently reported by Long COVID patients.
Gastric Dysbiosis and Microbiome Alterations
The gastric microbiome, whilst less diverse than the colonic microbiome, plays important roles in immune modulation and maintenance of mucosal integrity. Research demonstrates that SARS-CoV-2 infection disrupts gastric microbial composition, reducing beneficial commensal species whilst permitting overgrowth of potentially pathogenic organisms.
This dysbiosis extends beyond immediate infection risk. Altered gastric microbiota can:
- Promote chronic low-grade inflammation
- Impair nutrient bioavailability
- Disrupt gut-brain axis signalling
- Contribute to persistent gastrointestinal symptoms
The microbiome implications underscore why COVID-19 recovery often requires more than mere viral clearance—restoration of healthy microbial ecosystems is essential for complete physiological recovery.
Nutritional Implications: Vitamin B12 Deficiency and Beyond
Vitamin B12 Malabsorption Mechanism
Vitamin B12 (cobalamin) absorption is a complex, multi-stage process critically dependent on intrinsic factor. Dietary B12 binds to haptocorrin in the stomach, then transfers to intrinsic factor in the duodenum. The B12-intrinsic factor complex travels to the terminal ileum, where specific receptors facilitate absorption into the bloodstream.
When SARS-CoV-2 damages parietal cells, intrinsic factor production declines, creating a functional deficiency scenario similar to pernicious anaemia. Even with adequate dietary B12 intake, absorption becomes impaired, potentially leading to:
- Haematological effects: Megaloblastic anaemia, characterised by large, immature red blood cells and reduced oxygen-carrying capacity
- Neurological manifestations: Peripheral neuropathy, cognitive impairment, depression, and in severe cases, subacute combined degeneration of the spinal cord
- Metabolic consequences: Elevated homocysteine levels, associated with increased cardiovascular risk
Clinical Presentation in Post-COVID Patients

B12 deficiency symptoms often develop insidiously and may be attributed incorrectly to “Long COVID fatigue” without proper biochemical assessment. Common presentations include:
- Persistent fatigue and weakness
- Cognitive dysfunction (“brain fog”)
- Mood disturbances
- Paraesthesia (tingling/numbness in extremities)
- Balance difficulties
- Glossitis and oral lesions
These symptoms overlap substantially with reported Long COVID manifestations, suggesting that undiagnosed B12 deficiency may contribute to the persistent symptom burden in a subset of patients.
Broader Nutritional Vulnerabilities
Beyond B12, hypochlorhydria compromises absorption of other nutrients requiring acidic conditions:
- Iron: Gastric acid converts dietary ferric iron (Fe³⁺) to absorbable ferrous iron (Fe²⁺). Reduced acidity impairs this conversion, potentially causing iron deficiency anaemia.
- Calcium: Ionisation and solubilisation of calcium salts depend on gastric acidity, affecting bone health long-term.
- Magnesium: Similarly requires acidic conditions for optimal absorption.
- Protein digestion: Pepsinogen activation to pepsin (the primary gastric protease) requires low pH; impaired protein digestion may compromise amino acid availability.
Evidence-Based Nutritional Management Strategies
Assessment and Monitoring
Post-COVID patients, particularly those with persistent gastrointestinal symptoms, fatigue, or neurological complaints, warrant comprehensive nutritional assessment:
Laboratory investigations:
- Serum vitamin B12 (values >400 pmol/L optimal; <200 pmol/L clearly deficient)
- Methylmalonic acid (MMA) and homocysteine (more sensitive markers of functional B12 status)
- Complete blood count (assessing for macrocytic anaemia)
- Serum ferritin and iron studies
- Serum folate (B9), as deficiency can mask B12 deficiency haematologically
- Magnesium, calcium, and vitamin D status
Clinical assessment:
- Detailed dietary history
- Gastrointestinal symptom inventory
- Neurological examination if indicated
- Assessment of medication use (proton pump inhibitors, metformin, and other drugs affecting B12 absorption)
Therapeutic Interventions
Vitamin B12 Supplementation
Given that intrinsic factor deficiency impairs oral B12 absorption, therapeutic approaches must account for this:
Intramuscular/subcutaneous administration:
- Initial loading: 1000 mcg hydroxocobalamin or cyanocobalamin IM/SC daily for 1-2 weeks
- Maintenance: 1000 mcg monthly or as clinically indicated
- This bypasses the need for intrinsic factor entirely
High-dose oral supplementation:
- 1000-2000 mcg daily orally
- Relies on passive diffusion (approximately 1-2% absorption without intrinsic factor)
- Effective for many patients but requires adherence and monitoring
Sublingual/buccal preparations:
- May offer enhanced bioavailability compared to standard oral tablets
- Useful for patients preferring non-injectable options
Gastric Acid Support (When Appropriate)
For patients with documented hypochlorhydria and no contraindications:
- Betaine HCl supplementation: Taken with protein-containing meals under professional supervision
- Apple cider vinegar: Diluted in water before meals (traditional remedy with limited but supportive evidence)
- Digestive bitters: Stimulate endogenous acid production
Important caveat: These approaches are contraindicated in patients with active gastritis, peptic ulcers, or gastroesophageal reflux disease (GORD). Clinical judgement is essential.
Probiotic and Prebiotic Support
Emerging evidence supports targeted microbiome interventions post-COVID:
Probiotic strains with evidence:
- Lactobacillus and Bifidobacterium species for general gut health restoration
- Saccharomyces boulardii for H. pylori suppression (adjunct to antibiotic therapy if colonisation confirmed)
Prebiotic fibres:
- Inulin, fructo-oligosaccharides (FOS), and galacto-oligosaccharides (GOS)
- Support beneficial bacterial growth
- Found in foods like onions, garlic, leeks, asparagus, bananas, and whole grains
Fermented foods:
- Yoghourt, kefir, sauerkraut, kimchi, miso, tempeh
- Provide diverse microbial exposure and bioactive metabolites
Comprehensive Nutritional Optimisation
A whole-foods, anti-inflammatory dietary pattern supports recovery:
- High-quality protein: Supports tissue repair and immune function (1.2-1.5 g/kg body weight)
- Omega-3 fatty acids: Anti-inflammatory effects (fatty fish, flaxseed, walnuts)
- Antioxidant-rich foods: Berries, leafy greens, cruciferous vegetables
- Polyphenols: Green tea, dark chocolate, extra virgin olive oil
- Adequate hydration: Essential for all physiological processes
Screening and Prevention: Clinical Recommendations
Who Should Be Screened?
Post-COVID B12 screening is particularly important for:
- Patients with persistent fatigue, cognitive dysfunction, or neuropathy post-infection
- Individuals with pre-existing risk factors (vegetarian/vegan diet, age >60, metformin use, PPI therapy)
- Those with gastrointestinal symptoms lasting >3 months post-acute COVID
- Patients with known gastric pathology (chronic gastritis, previous H. pylori infection)
H. pylori Testing Considerations
Given increased colonisation risk post-COVID, consider H. pylori testing via:
- Urea breath test: Non-invasive, highly sensitive and specific
- Stool antigen test: Convenient, reliable
- Serology: Indicates exposure but not active infection
- Endoscopic biopsy: Gold standard but invasive; reserved for specific clinical indications
If H. pylori is detected, standard triple or quadruple therapy eradication protocols should be implemented, followed by confirmation of eradication 4-6 weeks post-treatment.
Long-Term Implications and Research Directions
Chronic Disease Risk
The long-term consequences of COVID-19-induced gastric dysfunction remain under investigation. Potential concerns include:
- Increased gastric cancer risk: Both hypochlorhydria and H. pylori colonisation are established risk factors
- Autoimmune gastritis development: Some evidence suggests SARS-CoV-2 may trigger autoimmune responses against parietal cells
- Persistent microbiome alterations: Unclear whether dysbiosis fully resolves or contributes to chronic inflammatory conditions
Emerging Therapeutic Approaches
Research is exploring novel interventions:
- Targeted probiotic formulations: Strains specifically selected for gastric microbiome restoration
- Postbiotic compounds: Microbial metabolites (short-chain fatty acids, peptides) with therapeutic potential
- Mucosal healing agents: Zinc carnosine, glutamine, and other compounds supporting gastric epithelial repair
Conclusion: Integrating Gastric Health into Post-COVID Care
The evidence is unequivocal: SARS-CoV-2 exerts significant, persistent effects on gastric physiology that extend far beyond the acute infection phase. Parietal cell destruction compromises both protective gastric acidity and nutritional absorption capacity, creating vulnerabilities to infection, microbiome disruption, and deficiency states particularly vitamin B12.
This gastric pathology fundamentally distinguishes COVID-19 from seasonal influenza and underscores why post-COVID care must adopt a comprehensive, systems-based approach. Clinical dietitians and healthcare professionals play a vital role in:
- Recognising gastric and nutritional complications as legitimate post-COVID sequelae
- Screening appropriately for B12 deficiency and H. pylori colonisation
- Implementing evidence-based nutritional interventions tailored to individual presentations
- Supporting microbiome restoration through diet, probiotics, and lifestyle modifications
- Monitoring long-term outcomes and adjusting strategies as recovery progresses
For patients navigating Long COVID, understanding these mechanisms empowers informed self-advocacy and collaborative care planning. Nutritional status is not peripheral to recovery it is foundational.
At Smart Nutrition International, we remain committed to translating emerging research into actionable clinical guidance, bridging the gap between scientific discovery and practical application for optimal patient outcomes.
References:
- Frontiers in Microbiology (2023). SARS-CoV-2 and gastric parietal cell damage. DOI: 10.3389/fmicb.2023.1177741
- PubMed (2025). COVID-19 gastric manifestations and vitamin B12 absorption. PMID: 40542875


