Published by Smart Nutrition International | Evidence-Based Clinical Nutrition
For decades, lactate has been dismissed as metabolic waste a cellular byproduct of intense exercise with little biological significance. Yet emerging research reveals a far more sophisticated story: this once-maligned molecule plays a pivotal dual role in cancer biology, simultaneously fuelling tumour growth and empowering our immune system’s anti-cancer defences. Understanding this paradox could unlock revolutionary therapeutic strategies for cancer patients worldwide.
The Dark Side: Lactate in the Tumour Microenvironment
Cancer cells possess a peculiar metabolic signature. Even with adequate oxygen supply, they preferentially convert glucose into lactate a phenomenon called the Warburg effect. This creates an acidic tumour microenvironment (TME) with lactate concentrations far exceeding normal tissues.
Traditionally, this elevated lactate benefits tumour progression through multiple mechanisms. Lactate contributes to disease progression through its multifaceted biological functions, serving as a central mediator in tumour immune evasion. The acidic environment impairs T-cell function, promotes blood vessel formation feeding tumour growth, facilitates cancer cell migration, and reduces treatment effectiveness.
Recent discoveries have identified lactylation a novel post-translational modification where lactate-derived groups attach to proteins, fundamentally altering gene expression. This metabolic-epigenetic crossroad influences immune cell polarisation, inflammatory responses, and therapeutic resistance, complicating the cancer treatment landscape.
The Remarkable Discovery: Lactate Fuels Anti-Tumour Immunity
Here’s where the narrative transforms dramatically. Groundbreaking research published in Cell Metabolism revealed that effector T lymphocytes immune cells responsible for attacking tumours utilise lactate as their primary mitochondrial energy source, even above glucose. This lactate fuels the Krebs cycle, substantially increasing energy production necessary for robust immune responses.
Research has shown that lactate, when separated from acidic protons, increases stemness of CD8+ T cells and augments anti-tumour immunity. These CD8+ cytotoxic T lymphocytes are the immune system’s primary assassins against cancer cells.
When separated from acidic protons, lactate inhibits histone deacetylase activity, increasing acetylation at specific gene locations. This enhances expression of genes promoting T cell stemness a phenotype characterized by self-renewal capacity, enhanced longevity, increased treatment responsiveness, and superior tumour-killing capacity.
Single-cell transcriptomics analysis revealed an increased proportion of stem-like TCF-1-expressing CD8+ T cells among intra-tumoural cells following lactate treatment, validating lactate’s direct immune-enhancing effects.
Exercise: Nature’s Immunotherapy Enhancement
The connection between exercise and improved cancer outcomes has been documented for years, but mechanisms remained elusive. Research demonstrated that exercise requires cytotoxic T cells to affect tumour growth, providing the missing link.
Studies showed that lactate and TCA metabolites accumulated in active skeletal muscle, plasma, and secondary lymphoid organs after acute exertion, exposing circulating immune cells to high levels of these metabolites.
When researchers administered lactate to mice with colon cancer or melanoma, remarkable results emerged. Tumour growth was significantly reduced compared to glucose-treated controls, and the anti-tumour effect was completely dependent on CD8+ T cells. When combined with immune checkpoint inhibitors, approximately 50% of mice became tumour-free. Daily infusions of sodium L-lactate at doses resulting in plasma lactate levels similar to intensive exercise showed decreased overall tumour growth.
Research suggests exercise, which naturally elevates lactate levels, may protect against cancer or augment the immune system’s cancer-fighting capacity. Daily doses of 2 g/kg sodium L-lactate producing lactate spikes similar to intensive exercise sessions showed significant anti-tumour effects with increased frequencies of both CD4+ and CD8+ T cells within tumours.
Clinical Implications: Targeting Lactate in Cancer Therapy
The dual nature of lactate presents both challenges and opportunities. Although lactate-targeted therapies show considerable promise, they remain in early developmental stages, requiring substantial efforts to unlock their full potential.
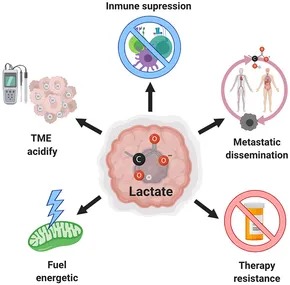
Promising Therapeutic Strategies
Lactate Production Inhibitors: LDH (lactate dehydrogenase) inhibitors target the Warburg effect directly, potentially reducing tumour lactate production whilst sensitising tumours to immunotherapy.
Lactate Transport Modulators: MCT1 and MCT4 (monocarboxylate transporter) inhibitors control lactate movement between cells, potentially preventing cancer cells from exporting lactate whilst allowing immune cells to import it.
pH Modification Strategies: Buffering the tumour microenvironment reduces acidity, preserving lactate’s beneficial immune effects whilst neutralising lactic acid’s immunosuppressive properties.
Lactate Supplementation Approaches: Controlled lactate administration boosts immune function, potentially timed with immunotherapy for synergistic effects and combined with exercise protocols.
Integration with Existing Immunotherapies
Research indicates CD8+ T cells pre-treated with lactate in vitro efficiently inhibit tumour growth upon adoptive transfer. This opens possibilities for enhanced CAR-T cell therapy, improved checkpoint inhibitor responses, cancer vaccine potentiation, and optimised adoptive cell transfer.
The Microbiota Connection: Another Layer of Complexity
The gut microbiota significantly influences cancer immunotherapy efficacy by modulating immune responses, remodelling the tumour microenvironment, and producing key metabolites.
The gut microbiota modulates cytotoxic CD8+ T cell function and adaptive anti-tumour immunity, with specific commensal strains capable of inducing interferon-gamma-positive CD8+ T cells to potentiate anti-cancer immunity.
Key mechanisms include short-chain fatty acid production (butyrate, propionate, acetate influencing T cell function), tryptophan metabolism affecting immune regulation, secondary bile acid synthesis influencing inflammation, and vitamin synthesis supporting immune function.
Strategies such as faecal microbiota transplantation, probiotics, and dietary interventions show promise in enhancing immunotherapy responses whilst reducing immune-related adverse events.
MY THOUGHTS: The Nutritional Nexus
As a clinical nutritionist specialising in the microbiota-cancer-immunity axis, I find these findings profoundly exciting yet humbling. What we’re witnessing is the convergence of three critical domains: metabolic reprogramming, immune function, and microbial ecology.
The lactate story exemplifies why reductionist thinking falls short in clinical nutrition. We cannot simply label lactate as “good” or “bad” context determines function. Similarly, the gut microbiome continuously communicates with our metabolism and immune system through metabolites like short-chain fatty acids, which share mechanistic similarities with lactate in their epigenetic effects.
From a clinical perspective, this research reinforces several key principles:
Individualised nutrition matters immensely. The same metabolite can promote or inhibit cancer depending on tumour microenvironment, immune status, and microbial composition. Precision nutrition approaches considering these variables will likely outperform one-size-fits-all interventions.
Lifestyle factors like exercise aren’t merely supportive—they’re potentially therapeutic. Exercise-induced lactate’s ability to enhance CD8+ T cell function suggests physical activity prescriptions should be integral to cancer care protocols, not afterthoughts.
The microbiota-metabolism-immunity triangle demands attention. Through dietary interventions, faecal microbial transplant, prebiotics, and probiotics, host anti-tumour immunity can be enhanced by promoting beneficial bacteria and their metabolites. Optimising gut health through targeted nutritional strategies may amplify both conventional and emerging cancer therapy benefits.
Timing and context are everything. Just as lactate’s effects depend on pH and cellular environment, nutritional interventions must be thoughtfully timed relative to treatment cycles to maximise benefit and minimise interference.
The future of oncology nutrition lies in understanding these complex interactions and translating them into actionable, evidence-based protocols supporting both treatment efficacy and quality of life.
Practical Takeaways
For Cancer Patients
- Maintain regular physical activity: Moderate to vigorous exercise may enhance immune function through lactate-mediated mechanisms
- Optimise gut health: A diverse, fibre-rich diet supports beneficial microbial populations
- Consider timing: Discuss with your oncology team whether exercise should be timed relative to immunotherapy treatments
- Avoid unnecessary antibiotics: Preserve microbiome diversity when possible
For Healthcare Practitioners
- Integrate exercise prescriptions: Include structured physical activity as comprehensive cancer care
- Support microbiome health: Implement dietary strategies promoting beneficial microbial populations
- Collaborate across specialities: Work with exercise physiologists, dietitians, and oncologists for integrated care
- Stay updated: This rapidly evolving field requires continuous education
Conclusion: Rethinking Lactate in Cancer Care
The journey from viewing lactate as metabolic waste to recognising it as a powerful immune modulator represents a fundamental shift in cancer biology. As we continue unravelling complex relationships between metabolism, immunity, and the microbiome, lactate stands as testament to biological sophistication a single molecule capable of vastly different effects depending on context.
The future of cancer treatment may include prescribed exercise protocols, carefully timed lactate supplementation, microbiome optimisation, and metabolically informed immunotherapy all working synergistically. For patients, practitioners, and researchers alike, the lactate story reminds us that sometimes the most powerful therapeutic tools are those the body already possesses; we simply need to understand how to leverage them effectively.

